Chapter 3 – Lithium Ion battery types
Note:
To read Chapter 1, please visit: https://www.norwatt.es/noticia-industrial-batteries--features-summary---chapter-1---acronyms-es.html
To read Chapter 2, please visit: https://www.norwatt.es/noticia-industrial-batteries--features-summary--chapter-2---industrial-battery-types-es.html
____________________________________________________________
Commercially, there are five types of lithium-ion batteries depending on the cathode material and another two depending on the material of the anode material.
Working Principles
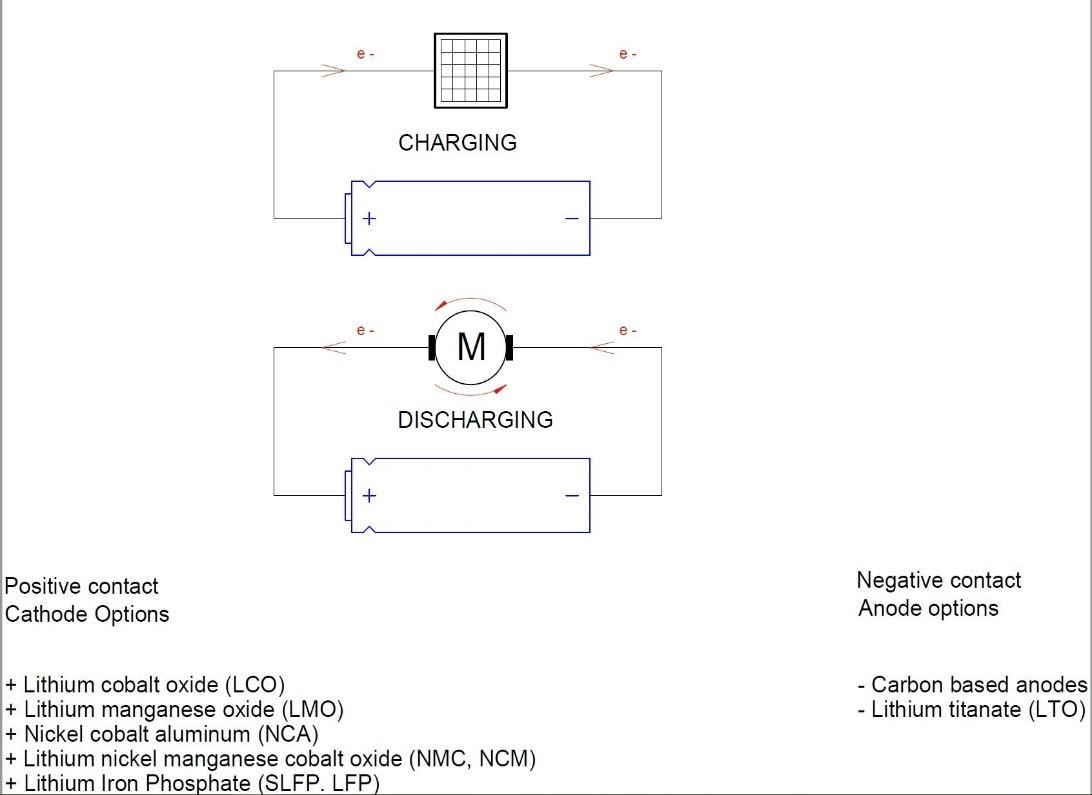
Cells convert chemical energy into electrical energy (and vice-versa). In Lithium ion batteries, during cell charge, the lithium ions flow from the cathode to the anode and they are stored there. During cell discharge, lithium ions disassociate from the anode and flow to the cathode through the electrolyte and electrons are transported by the external circuit to do an effective work.
A lithium ion battery has three functional layers: the positive electrode (cathode), the negative electrode (anode), and the separator.
The anode corresponds to the negative electrode. The electrons leave the battery through the anode; therefore, it must be made of a material with high electronic conductivity and great cycling capacity. Graphite is generally used. (70 to 250 micron)
The cathode corresponds to the positive electrode and must be capable of accepting and releasing lithium ions and electrons. Cathodes are generally made of metallic lithium oxide which can oxidize when lithium is removed. For example: LiMO2, (M = cobalt (Co), nickel (Ni)####manganese (Mn)). (70 to 250 micron)
The mode of transporting lithium ions between electrodes during charging and discharging is the electrolyte; that’s why it has to be a good ionic conductor and a good electronic insulator. The electrolytes are usually made up of a solution of lithium salt in an organic solvent; for example: lithium hexafluorophosphate (LiPF6) with ethylene carbonate and methyl carbonate (EC-DMC).
The membrane that separates the anode and the cathode is called the separator and it is the host of the electrolyte. The separator prevents short circuits between anode and cathode but allows the Li ions to flow during charging and discharging. (20 to 25 micron)
Batteries can be classified by energy and power. This classification is marked by the composition and characteristics of the electrodes and their materials: Energy for long time discharge and power for high current discharge.
High Power cells – can discharge at rates 10 to 100C rate (from 6 min to
High Energy cells – can discharge at 2C rate (30 min####less but energy content exceeds 170 to 190 Wh/kg
Energy storage capacity of a battery: depends on the number of active components that the electrodes can store.
Power capacity: is a function of the surface area of the electrodes and the internal resistance of the battery.
These energy and power ratings depend on and are related to each other. The rate-C represents this energy / power ratio (it is the data that allows knowing how a battery is charge####discharged in relation to its maximum capacity). It should be added that the energy density depends on the chemistry of the battery, while the power density depends on the kinetics and the design of the cell. Thin electrodes can provide high power densities, and thick electrodes produce high energy densities. This is why batteries can be designed to supply high powe####high energy depending on the needs.
3.1 POSITIVE CONTACT: CATHOTE OPTIONS
The positive electrode is called the cathode and typically defines the chemical type of the battery. It is built with an aluminium full coated in both sides with a lithium metal oxide compound and binder
Generally, research into lithium ion batteries is being carried out to find an economical material, with better cyclic performance, good thermal stability and conductive to high energy and power densities. In particular, thermal stability is a major concern when######ing cathode materials since heat generation at the cathode is three to four times greater than that of the anode.
3.1.1 Lithium cobalt oxide (LCO) (LiCoO2)
LCO has high energy density per unity mass, but is not well suited for use in off-grid products because of lower cycle life and poorer safety. Nonetheless, LCO batteries are ubiquitous and still frequently used for these applications.
|
Characteristic |
LiCoO2 (LCO) |
|
Nominal voltage per cell |
3,7 Vdc (4,2 Vdc 100% SOC) |
|
Specific energy (Wh/kg) |
175-200 |
|
Energy density (Wh/L) |
400-640 |
|
Cycle life (to 80% original capacity at 100% DOD) |
500 |
|
Calendar life (years) |
>5 |
|
Ambient temperature during charge (°C) |
0-45 |
|
Ambient temperature during discharge (°C) |
-20-60 |
|
Self-discharge capacity loss per month |
2-10% |
|
Memory effect |
No |
|
Toxic metals |
No |
|
Battery management system required |
Yes |
Main disadvantages:
The Li-cobalt is losing favour to Li-manganese, but especially NMC and NCA because of the high cost of cobalt and improved performance by blending with other active cathode materials.
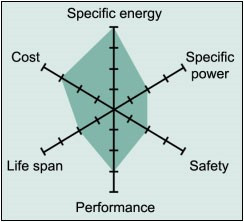
Li-cobalt excels on high specific energy but offers only moderate performance specific power, safety and life span.
LCO technology LiCoO2 available here, please check:
https://www.norwatt.es/products/batteries-and-accessories-lithium-ion-batteries-saft-sally-en.html
3.1.2 Lithium manganese oxide (LMO) (LiMn2O4)
Pure LMO batteries have good thermal stability and safety but lower cycle life; they have been replaced by blending the manganese oxide with nickel and cobalt (NMC).
|
Characteristic |
LiMn2O4 (LMO) |
|
Nominal voltage per cell |
3.70V (3.80V) nominal; typical operating range 3.0–4.2V/cell |
|
Specific energy (Wh/kg) |
100-150 |
|
Energy density (Wh/L) |
150-300 |
|
Cycle life (to 80% original capacity at 100% DOD) |
300-700 |
|
Calendar life (years) |
>5 |
|
Ambient temperature during charge (°C) |
5-45 |
|
Ambient temperature during discharge (°C) |
5-45 |
|
Self-discharge capacity loss per month |
--- |
|
Memory effect |
No |
|
Toxic metals |
No |
|
Battery management system required |
Yes |
Main advantages:
Main disadvantages:
Li-manganese has a capacity that is roughly one-third lower than Li-cobalt. Design flexibility allows engineers to maximize the battery for either optimal longevity (life span), maximum load current (specific power) or high capacity (specific energy).
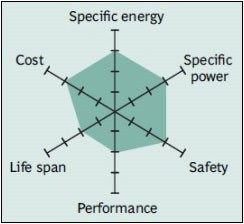
Although moderate in overall performance, newer designs of Li-manganese offer improvements in specific power, safety and life span.
https://www.norwatt.es/archivos/filemanager/2020/06/15_ii427cbrjm_Chapter_3_para_FAQ.pdf
LinkedIn:
Rodrigo Suárez - CEO: https://es.linkedin.com/in/rodrigo-suarez-cueto-05ab9357
NORWATT SL:https://cl.linkedin.com/company/norwatt-sl