Chapter 3 – Lithium Ion battery types
Note:
To read Chapter 1, please visit: https://www.norwatt.es/noticia-industrial-batteries--features-summary---chapter-1---acronyms-es.html
To read Chapter 2, please visit: https://www.norwatt.es/noticia-industrial-batteries--features-summary--chapter-2---industrial-battery-types-es.html
_____________________________________________________________________________________________________________________________
3.1.4 Lithium nickel manganese cobalt oxide LiNiMnCoO2 (NMC)
One of the most successful Li-ion systems is a cathode combination of nickel-manganese-cobalt (NMC). The secret of NMC lies in combining nickel and manganese.
NMC blends offer combinations of good cycle life, safety, and high energy density. The ratios of elements can be tailored to emphasize qualities that target specific applications including off-grid products.
Battery manufacturers move away from cobalt systems toward nickel cathodes because of the high cost of cobalt. Nickel-based systems have higher energy density, lower cost, and longer cycle life than the cobalt-based cells but they have a slightly lower voltage.
NMC batteries are widely used for power tools, energy storage systems (EES) which need frequent cycling and electrical power-trains like automotives. The cathode combination typically used is 1-1-1 which means one third nickel, one third manganese and one third cobalt. This reduces the raw material cost due to lowered content of cobalt. Other combinations are also possible.
|
Characteristic |
LiNiMnCoO2 (NMC) |
|
Nominal voltage per cell |
3.60V, 3.70V nominal; typical operating range 3.0–4.2V/cell####higher |
|
Specific energy (Wh/kg) |
150-220 |
|
Energy density (Wh/L) |
205-300 |
|
Cycle life (to 80% original capacity at 100% DOD) |
1000-2000 |
|
Calendar life (years) |
>10 |
|
Ambient temperature during charge (°C) |
-10 + 45 |
|
Ambient temperature during discharge (°C) |
-10 +45 |
|
Self-discharge capacity loss per month |
0,35 – 2,5 |
|
Memory effect |
No |
|
Toxic metals |
Yes |
|
Battery management system required |
Yes |
Main advantages:
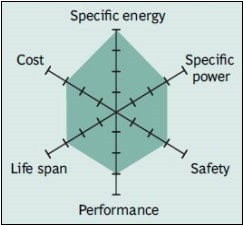
NMC has good overall performance and excels on specific energy. This battery is the preferred candidate for the electric vehicle and has the lowest self-heating rate.
NMC technology LiNiMnCoO2 available here, please check:

3.1.5 Lithium Iron Phosphate (SLFP, LFP) LiFePO4
LiFePO4 batteries exhibit qualities that make them ideally suited for off-grid products where cost, safety, stability, and cycle life are primary requirements. It does not release oxygen, which makes it a safe material to use with better thermal stability.
LFP is more tolerant to full charge conditions and is less stressed than other lithium-ion systems if kept at high voltage for a prolonged time. As a trade-off, its lower nominal voltage of 3.2V/cell reduces the specific energy below that of cobalt-blended lithium-ion. With most batteries, cold temperature reduces performance and elevated storage temperature shortens the service life, and Li-phosphate is no exception. Li-phosphate has a higher self-discharge than other Li-ion batteries, which can cause balancing issues with aging.
The LiFePO4 batteries can improve electrochemical performance is to nano-structure LiFePO4 particles or coating the cathode with Sn.
|
Characteristic |
LiFePO4 (LFP) |
|
Nominal voltage per cell |
3.20, 3.30V nominal; typical operating range 2.5–3.65V/cell |
|
Specific energy (Wh/kg) |
90-120 |
|
Energy density (Wh/L) |
125-250 |
|
Cycle life (to 80% original capacity at 100% DOD) |
1000 – 2000 (can retain 95% capacity after 1000 cycles) |
|
Calendar life (years) |
>5 |
|
Ambient temperature during charge (°C) |
0-45 |
|
Ambient temperature during discharge (°C) |
-30-60 |
|
Self-discharge capacity loss per month |
2-10 |
|
Memory effect |
No |
|
Toxic metals |
No |
|
Battery management system required |
Yes |
Main advantages:
Main disadvantages:
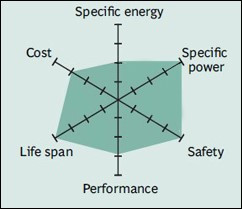
Li-phosphate has excellent safety and long life span but moderate specific energy and elevated self-discharge.
SLFP technology LiFePO4 available here, please check:

https://www.norwatt.es/products/batteries-and-accessories-lithium-ion-batteries-saft-flexion-en.html
3.2 NEGATIVE CONTACT: ANODE OPTIONS
Anode composition and electrochemical properties are very important to the performance of a battery. For the anode material to be effective, high electronic conductivity, low working potential, high cycle stability and low volume change are required during lithium######ion and extraction.
In order to be suitable for lithium-ion battery manufacturing, anode materials should meet the following requirements:
During initial Lithium ion battery operation, the SEI (solid electrolyte interphase) layer forms on the graphite surfaces, the most common anode material. The SEI is essential to the long-term performance of LIBs, and it also has an impact on its initial capacity loss, self-discharge characteristics, rate capability, and safety. While the presence of the anode SEI is vital, it is difficult to control its formation and growth, as they depend on several factors. These factors include the type of graphite, electrolyte composition, electrochemical conditions, and temperature.
3.2.1 Carbon based anodes
Graphite formulations (A cooper foil coated with natural graphite, synthetic graphite or amorphous carbon) are used for the negative electrode in the majority of Li-ion commercial cells.
Graphite and carbon-based anode materials have been used in commercial Li-ion cells since 1991 because of the good electrochemical properties, low cost, and nontoxicity of graphite.
During the first charge cycle a solid electrolyte interphase layer (SEI) forms in the graphite surface that helps to stabilize the molecular structure and prevent it from reaching with the liquid electrolyte.
The SEI layer stabilizes the anode by preventing reactions between the graphite and the electrolyte. SEI layer integrity plays an important role in cell performance. The SEI can therefore be described as a protective layer.
The secondary SEI mainly consists of Li2CO3 and LiF. It has been suggested that besides the formation of the secondary SEI, a new organic SEI is formed by solvent reduction. These complex processes of SEI formation and change occur up to a temperature of approx. 200°C.
3.2.2 LTO cells
The LTO (Li4Ti5O12) is bringing a new dimension of possibilities for the energy storage with a number of economical as well as ecological aspects.
With applications in many sectors, with a primary focus on high speed charging and energy storage, LTO technology is the future of battery-powered technology.
LTO cells offers very high cycle life, excellent thermal stability, excellent safety, and good low temperature operation. LTO can be used with NMC or LMO cathodes.
LTO does not react with organic electrolytes, therefore SEI is not formed which makes it highly thermal stable and an excellent choice